electroluminescence (EL)
Electroluminescence is a phenomenon in which certain materials emit light when an electric field is applied. The material is excited by the electric field lines, which causes electrons in compound semiconductors to jump the band gap and change from one conductive band to another, emitting light.
There are various electronic components that emit light. These include light-emitting diodes(LEDs), electroluminescent (EL) films and OLEDs.
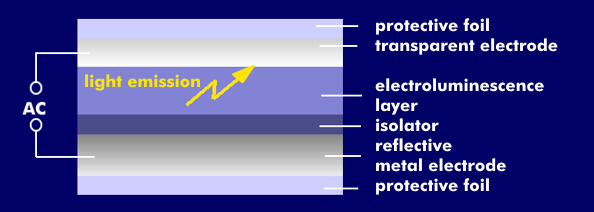
As for electroluminescent (EL) film, it is an approximately 0.8 mm thin multilayer film with transparent and reflective electrodes between which the doped and insulated light-emitting electroluminescent layer is located. It consists of a II-VI compound semiconductor such as zinc sulfide doped with silver, gold, gallium or copper. The transparent electrode is made of indium tin oxide( ITO), and the reflective electrode is made of a conductive metal foil. The electrodes are protected by outer plastic foils.
As soon as voltage is applied to the electrodes, an electric field is built up, which excites the EL layer to emit light. The electric field raises free electrons to a higher energy level. When they fall back to their ground level, they emit light, the emitted wavelength being material dependent. The alternating voltage applied to the electrodes ensures that they fall back to their base level. The basic colors emitted are white, light blue, light green, blue-green and orange. By changing the frequency, the colors can be changed in certain ranges.
The AC voltage is between 20 V and 200 V and has a frequency between 100 Hz and 2 kHz. The frequency affects the luminous color. The luminous efficiency and the luminance are relatively low. The luminous efficiency is max. 10 lm/W and the luminance is about 20 cd/m2 to 200 cd/m2. Power consumption is in the milliwatt range at about 20 mW/cm2. The light produced by luminescent foils is monochromatic, and the color temperature depends on the frequency, band gap and supply voltage.