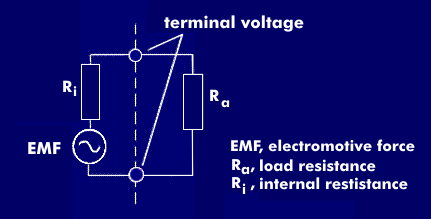
terminal voltage
Terminal voltage is the voltage applied to battery terminals and is dependent on various voltage drops.
The terminal voltage results from the open-circuit voltage of the battery, which depends on the state of charge of the battery, the temperature and the concentration of the electrolyte. If no current flows, the open-circuit voltage corresponds to the electromotive force( EMF).
When current flows, the open-circuit voltage is reduced by the voltage drop across the internal resistance, which is determined by lead losses, contact resistances, and electrode conductivity. Further voltage drops result from the electrochemical reaction and from reactances at the reaction plates. The sum of these voltage drops reduces the open-circuit voltage and gives the terminal voltage.