redox flow battery (RFB)
A redox flow battery is a rechargeable energy storage device, a secondary cell for medium and higher power applications. The term redox is composed of the first letters of reduction and oxidation. Reduction stands for electron uptake and oxidation for electron release. The principle of the redox flow battery is based on the redox reaction.
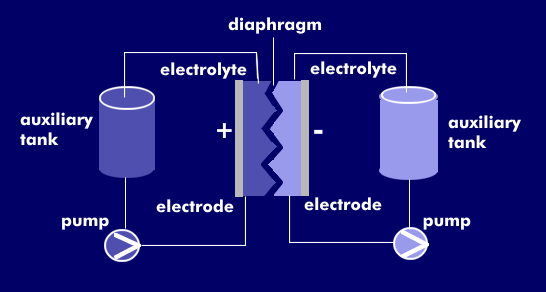
The redox flow battery is a liquid battery or wet cell based on chemical conversion and uses two separate electrolyte reservoirs. The energy is stored in the electrolytes. The electrolyte is pumped through the two electrolyte reservoirs and circulates past the membrane, which separates the two electrolytes and prevents them from mixing. Ion exchange occurs across the membrane, releasing energy through reduction or oxidation. This energy can be tapped as the voltage difference between the electrodes, with which each electrolyte container is equipped.
Redox flow batteries can be used as quarter storage ofphotovoltaic systems, in home storage systems or as buffer batteries in power supply and UPS systems. Their power ranges from a few hundred watts to several megawatts. The energy density is not very high and depends on the electrolyte. Depending on the electrolyte, it is below or slightly above the energy density of lead batteries. Vanadium, which is often used in the electrolyte solution, has an energy density of 25 Wh per liter, while sodium bromide is about twice as high in energy density. Furthermore, solutions of chromium, titanium and sulfur are used as electrolytes.
The amount of electrolyte is decisive for the storage capacity. This means that the size of the electrolyte container determines the storage capacity. The efficiency of redox flow batteries is very high, as is the number of charging cycles, which is over 10,000. Self-discharge does not take place.