proton exchange membrane fuel cell (PEMFC)
Fuel cells are divided into low-temperature fuel cells and high-temperature fuel cells. The PEM fuel cell, Proton Exchange Membrane Fuel Cell (PEMFC) or Polymer Electrolyte Fuel Cell (PEFC), belongs to the former group, and the SO fuel cell, Solid Oxide Fuel Cell( SOFC), to the latter.
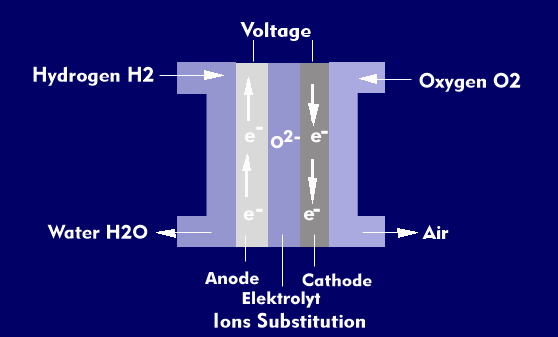
In the PEM fuel cell, chemical energy is converted into electrical energy using hydrogen (H2) and oxygen (O2). The process is the reverse of electrolysis.
The PEM fuel cell consists of the anode and the cathode, which is coated with platinum or a platinum compound such as platinum- nickel or platinum- cobalt. The platinum serves as a catalyst, without which hydrogen and oxygen cannot react. Between the two electrodes is a thin polymer membrane made of ceramic material, the proton exchange membrane (PEM), which acts as the central component. It is permeable to hydrogen ions (protons), but not to the electrons that have been removed from the hydrogen atoms. The protons react with oxygen at the cathode to form water.
In the electrochemical reaction, the oxidation reaction at the anode produces protons and electrons. As for the reduction reaction at the cathode, protons and electrons are combined there with oxidizing agents to form water. In the chemical reaction, the polymer membrane plays the crucial role for proton exchange because it conducts protons from the anode to the cathode. The proton exchange membrane also serves as a separator to separate the anode and cathode reactants in fuel cells and electrolyzers.
The operating temperature of the PEM fuel cell is between 60 °C and 120 °C, and the efficiency is about 60 %. The waste heat is between 60 °C and 80 °C. PEM fuel cells can generate electrical power from 0.3 W up to 500 kW. PEM fuel cells are suitable for use in electric vehicles, in buses, trucks, streetcars and ships.