emission layer (OLED) (EML)
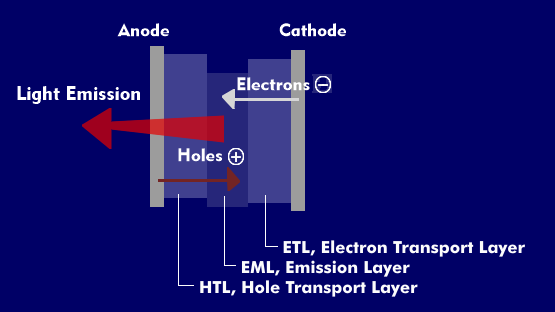
The light emission of OLEDs is based on electroluminescence. When a forward voltage is applied to an OLED, spurious electrons (holes) and electrons are injected as charge carriers. These injected charge carriers have to overcome the level barriers.
After overcoming the level barriers, the holes occupy the highest energy levels to be occupied of the Hole Transport Layer( HTL), the Highest Occupied Molecular Orbital (HOMO), and the electrons occupy the lowest unoccupied energy levels, the Lowest Unoccupied Molecular Orbital (LUMO), of the Electron Transport Layer( ETL). HOMO is similar to the valence band of semiconductors and LUMO is similar to the conduction band.
Due to the externally applied electric field, the injected holes, which have a positive charge, travel from the anode to the emission layer( EML) via the HTL layer, while the electrons also travel from the cathode to the EML layer via the ETL layer, where they recombine to form an electron-hole pair, an exciton, thereby emitting light in the indium tin oxide( ITO) coated anode, which is formed from a glass substrate.