doping
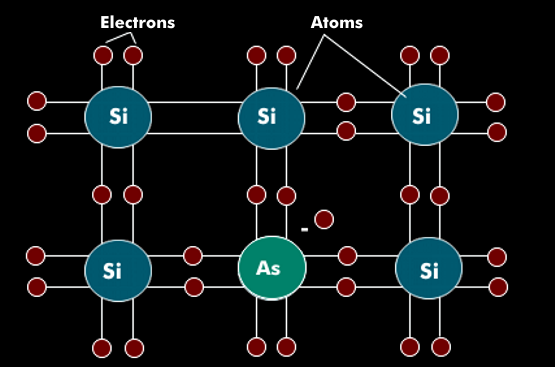
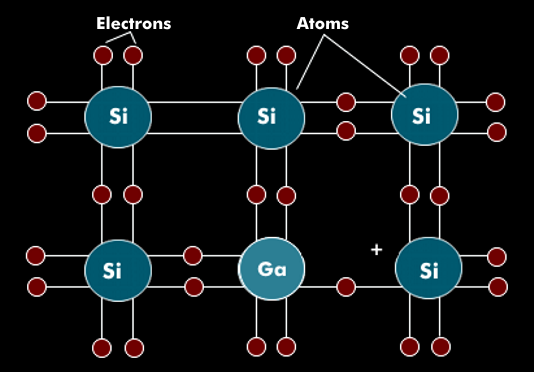
Doping is the process of contaminating high-purity semiconductor material with impurities whose chemical valence is different from that of the semiconductor material. The impurity atoms are inserted into the crystalline atomic semiconductor structure by ion bombardment or diffusion. If the inserted impurity atoms have more electrons in their valence band than the semiconductor atom, they are called donors or dopant atoms.
An inserted donor has excess electrons that cannot be incorporated into the crystalline semiconductor structure and are available for current transport. These electrons are called majority charge carriers and occur in n-doping; in p-doping, the holes are the majority charge carriers.
Phosphorus (P), arsenic( As) and antimony (Sb) are used as donors for silicon. In tetravalent silicon, the four bonds are occupied by pentavalent substances, with one charge carrier unbound. This doping with excess electrons is an n-doping or n- conductor.
If the inserted foreign atoms lack an electron in the valence band, then they are called acceptors, whose missing electrons leave missing electrons or holes. Aluminum (Al), gallium( Ga), indium (In) and boron (B) are used as acceptors for silicon. This doping with trivalent acceptors results in a missing bond; a positive hole is created that can be occupied by another electron. This doping with spurious electrons is a p-doping or a p-conductor. The excess of false electrons, or positive charge carriers, also serves to transport current. In silicon, boron (B) is usually used as an acceptor.
The concentration of impurity atoms is the doping strength, it determines the conductivity of the semiconductor material. It is extremely low and is in the range of `10^-4` to `10^-10` impurity atoms to one silicon atom. The higher the doping strength, the greater the conductivity of the doped material.